The JCCP is committed to raising public awareness of the risks associated with various non-surgical and hair restoration treatments. As part of this process the JCCP will publish regular ‘Information Sheets’ in key areas.
The Procedures that are Regulated by the JCCP – Introduction to non-surgical aesthetic treatments and hair restoration surgery
There has been a huge growth in non-surgical treatments and hair restoration surgery in recent years. For many members of the public this leads to great confusion. The JCCP has published a series of information sheets to give a brief explanation of each procedure and the risks involved.
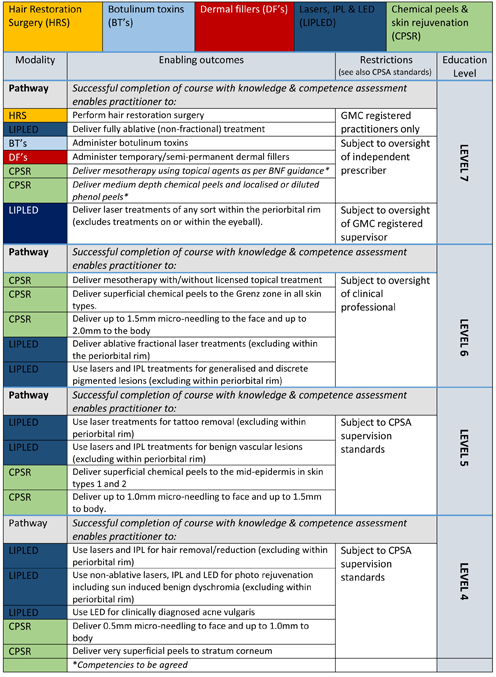
The JCCP currently recognises the following five procedural modalities that are performed by our Registrant Practitioners:
These procedural modalities are simply the treatments that we regulate currently according to a set of prescribed practice standards (as determined by the Cosmetic Practice Standards Authority) and formal educational qualifications that are set down by the JCCP.
In 2013 Sir Bruce Keogh presented his Government commissioned report into the cosmetic/aesthetic industry, which outlined a range of failings that present a risk to patients and service users. In response, the Government commissioned Health Education England (HEE) in 2013 to work with industry stakeholders and professional regulators to develop a framework of agreed qualifications and standards, to address practitioner failings, to improve public protection and to mitigate risks that might occur as the result of harmful practice.
Under the umbrella of HEE, multiple industry stakeholders and experts:
- Identified the modalities which were known to cause greatest concern and where risk is highest in relation to patient harm/safety.
- Developed a qualifications framework for the five modalities noted above which set out the education and practice requirements necessary to undertake cosmetic medicine safely.
- Defined the requirements for clinical oversight, paying attention to the role of the statutory regulated professional and, in particular, those professionals with a license to prescribe.
- Agreed for the need (in the absence of statutory regulation) for an over-arching authority – the JCCP and its sister organisation the CPSA - which would further develop these standards and oversee their implementation, under a mechanism of voluntary self-regulation.
Whilst 5 modalities are currently recognised by both the JCCP and by the CPSA, these are likely to increase in number in the future as new risks to person/patients are identified. It is the specific role of the CPSA to review new treatments as they emerge, to assess risk and where necessary to develop new standards.
Your JCCP registered practitioner
For each of the modalities your JCCP practitioner is registered to provide, you can be sure that they:
- Are qualified at the appropriate educational level.
- Are permitted to practise independently or otherwise subject to a clear process of delegation with prescribed oversight/supervision.
- Practise safely and ethically according to a robust set of standards/code of practice.
- Are subject to stringent fitness to practice rules where standards are not met.
Things to consider for all treatments
- Discuss with your practitioner and allow yourself time for a ‘cooling-off’ period. It is important to take enough time to consider all the information before making a decision to have treatment.
- Be thorough and honest in providing your medical history. Your practitioner requires these details not only to make a decision about your treatment, but to address problems – which often require prescription medicines - should they arise.
- Your consent to treatment should be ‘informed’. This means that you have been given all the relevant facts, both generally and as they relate specifically to you, and that you understand them. You should have the opportunity to ask questions and consider treatment options.
- If you do have a problem or concern after your treatment, the first step is always to contact your practitioner. They are best placed to advise, and to manage any side-effects. They should inform you how they can be contacted at
any time.
Treatment Modality Table